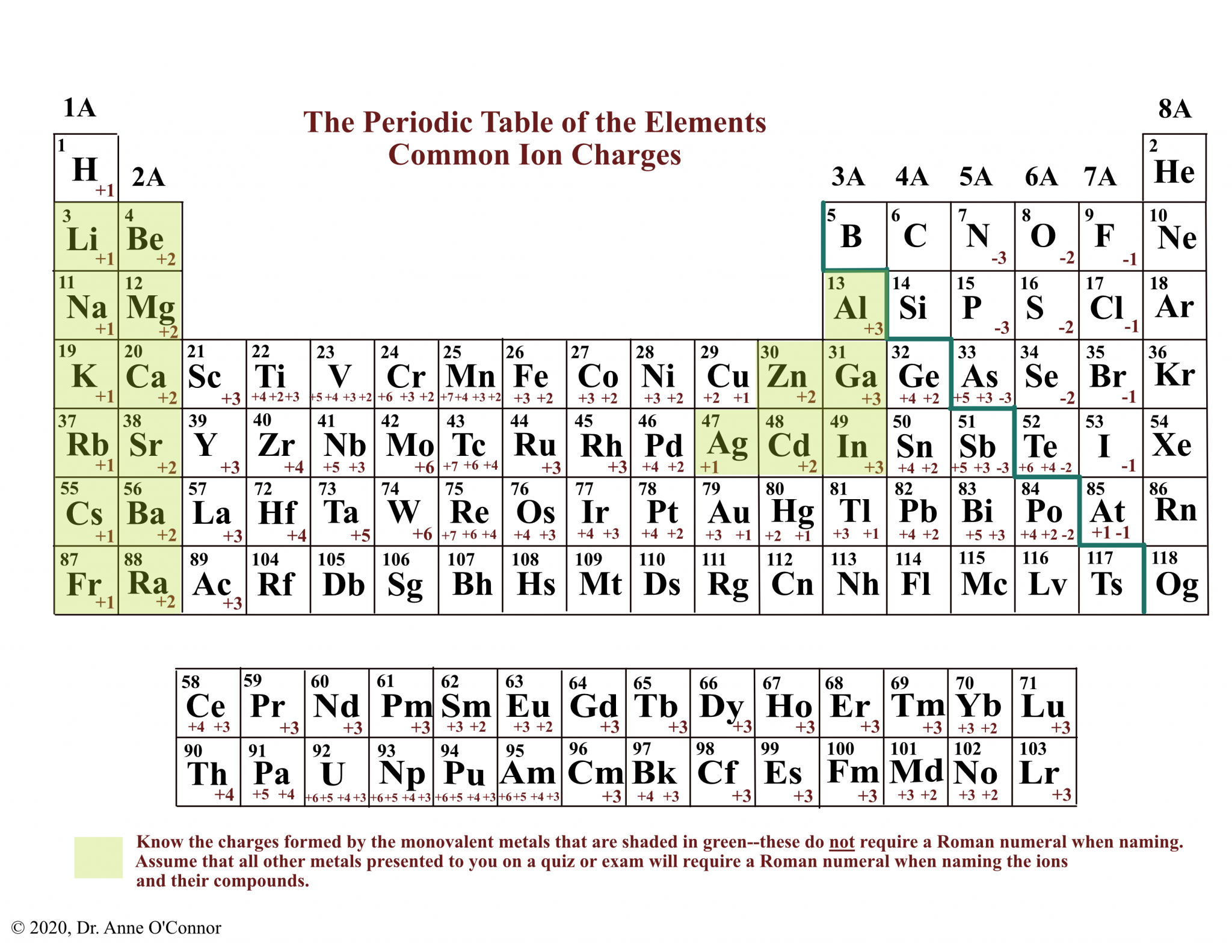
The Periodic Table is a fundamental tool in chemistry, organizing elements base on their atomic turn, electron form, and recur chemical properties. One of the key aspects of the Periodic Table is realize the Periodic Table Ionic Charges, which are all-important for forebode the behaviour of elements in chemical reactions. Ionic charges refer to the electric charge that an atom acquires when it gains or loses electrons to form ions. This concept is essential for understanding the constitution of ionic compounds and the properties of elements across the Periodic Table.
Understanding Ionic Charges
Ionic charges are determined by the routine of electrons an atom gains or loses to achieve a stable electron contour, typically that of a noble gas. Elements on the left side of the Periodic Table tend to lose electrons and form plus ions (cations), while elements on the right side tend to gain electrons and form negative ions (anions).
Determining Ionic Charges
To determine the ionic charge of an element, you need to cognise its perspective on the Periodic Table. Here are some general rules:
- Group 1 (Alkali Metals): These elements have one electron in their outermost shell and tend to lose this electron to form ions with a 1 charge. Examples include sodium (Na ) and potassium (K ).
- Group 2 (Alkaline Earth Metals): These elements have two electrons in their outermost shell and tend to lose these electrons to form ions with a 2 charge. Examples include magnesium (Mg 2 ) and calcium (Ca2 ).
- Group 17 (Halogens): These elements have seven electrons in their outermost shell and tend to gain one electron to form ions with a 1 charge. Examples include chlorine (Cl ) and fluorine (F ).
- Group 16 (Chalcogens): These elements have six electrons in their outermost shell and tend to gain two electrons to form ions with a 2 charge. Examples include oxygen (O 2 ) and sulfur (S2 ).
Transition Metals and Variable Ionic Charges
Transition metals, located in the middle of the Periodic Table, frequently exhibit variable ionic charges. This is because they have multiple electrons in their outermost shell that can be involved in adhere. for instance, iron (Fe) can form ions with charges of 2 (Fe 2 ) and +3 (Fe3 ). The specific charge depends on the chemical environment and the compound being formed.
Periodic Table Ionic Charges and Chemical Bonding
Understanding Periodic Table Ionic Charges is crucial for predicting the types of chemic bonds that will form between elements. Ionic bonds occur when one element loses electrons to another, leave in the establishment of ions that are attracted to each other due to their opposite charges. for instance, sodium (Na) and chlorine (Cl) form an ionic bond to create sodium chloride (NaCl), where sodium loses an electron to become Na and chlorine gains an electron to become Cl .
Predicting Ionic Charges from Electron Configuration
To predict the ionic charge of an element, you can seem at its electron shape. Elements tend to gain or lose electrons to achieve a stable electron configuration, which is typically that of the nearest noble gas. for representative, potassium (K) has the electron shape [Ar] 4s 1. To achieve a stable configuration like argon (Ar), potassium loses its single 4s electron to form K .
Common Ionic Charges of Elements
Here is a table of mutual ionic charges for some elements based on their position in the Periodic Table:
| Element | Group | Ionic Charge |
|---|---|---|
| Lithium (Li) | 1 | 1 |
| Beryllium (Be) | 2 | 2 |
| Sodium (Na) | 1 | 1 |
| Magnesium (Mg) | 2 | 2 |
| Aluminum (Al) | 13 | 3 |
| Chlorine (Cl) | 17 | 1 |
| Oxygen (O) | 16 | 2 |
| Sulfur (S) | 16 | 2 |
| Iron (Fe) | 8 | 2, 3 |
Note: The ionic charges name are the most mutual for these elements, but some elements, especially transition metals, can have multiple ionic charges depending on the compound.
Applications of Ionic Charges
Understanding Periodic Table Ionic Charges has numerous applications in chemistry and industry. for example, cognise the ionic charges of elements helps in predicting the solvability of compounds in water. Ionic compounds with extremely charge ions tend to be more soluble in h2o due to the strong attraction between the ions and h2o molecules.
In materials science, ionic charges are crucial for plan new materials with specific properties. For instance, the development of batteries relies on realise the ionic charges of the materials used in the electrodes and electrolytes. The movement of ions between the electrodes during accuse and discharge is what allows batteries to store and release energy.
Challenges in Determining Ionic Charges
While the general rules for set ionic charges are straightforward, there are challenges, particularly with conversion metals and elements in the p block. These elements can exhibit multiple oxidation states, do it difficult to predict their ionic charges without extra information. for instance, manganese (Mn) can have oxidation states ranging from 2 to 7, depending on the compound.
Another challenge is understanding the demeanour of elements in different chemical environments. The ionic charge of an element can modify based on the other elements it is bond to and the overall structure of the compound. For example, in some compounds, sulfur can have a charge of 2, while in others, it can have a charge of 4 or 6.
To overcome these challenges, chemists often use experimental techniques such as X ray crystallography and spectroscopy to determine the exact ionic charges of elements in a compound. These techniques provide detail information about the electronic structure and bonding in the compound, countenance for more accurate predictions of its properties.
besides experimental techniques, computational methods are also used to predict ionic charges. These methods involve using complex algorithms and models to simulate the behavior of atoms and molecules. While these methods are not always perfect, they can provide worthful insights into the properties of compounds and help guide data-based enquiry.
One of the most powerful computational methods for predicting ionic charges is concentration functional theory (DFT). DFT is a quantum mechanical method that calculates the electronic structure of atoms and molecules. By using DFT, chemists can predict the ionic charges of elements in a compound and read how these charges touch the compound's properties.
Another significant computational method is molecular dynamics simulations. These simulations model the movement of atoms and molecules over time, allowing chemists to study the dynamical behavior of compounds. By using molecular dynamics simulations, chemists can predict how ionic charges affect the constancy and reactivity of compounds.
While computational methods are powerful tools for predicting ionic charges, they also have limitations. These methods often rely on approximations and assumptions, which can lead to inaccuracies in the predictions. Therefore, it is important to use a combination of data-based and computational techniques to gain a comprehensive translate of ionic charges and their effects on chemic properties.
In compendious, interpret Periodic Table Ionic Charges is indispensable for foretell the doings of elements in chemic reactions and designing new materials with specific properties. While there are challenges in mold ionic charges, especially with transition metals and elements in the p block, a combination of observational and computational techniques can furnish worthful insights into the properties of compounds.
to summarize, the concept of Periodic Table Ionic Charges is profound to chemistry and has wide ranging applications in respective fields. By understanding how elements gain or lose electrons to form ions, chemists can predict the types of chemical bonds that will form, the solubility of compounds, and the properties of new materials. This knowledge is crucial for advancing our understanding of chemistry and develop new technologies that improve our lives.
Related Terms:
- charge ions on occasional table
- ion charge chart occasional table
- ion chart pdf
- periodic table of ion charges
- list of ions and charges
- ionic compounds list with charge