Understanding the electron arrangement of elements is fundamental to grasping their chemic properties and behaviors. Among the elements, sodium stands out due to its unparalleled position in the occasional table and its widespread use in assorted applications. This blog post delves into the electron arrangement of sodium, its significance, and how it influences sodium's chemic and physical properties.
What is Electron Arrangement?
Electron arrangement, also known as electron configuration, refers to the dispersion of electrons in the orbitals of an atom. This arrangement is crucial because it determines how an atom will interact with other atoms, organise bonds and participating in chemical reactions. The electron arrangement is typically symbolise using a notation that specifies the figure of electrons in each orbital.
The Periodic Table and Sodium
Sodium (Na) is an alkali metallic locate in Group 1 (IA) of the occasional table. It has the atomic turn 11, which means it has 11 protons and 11 electrons. The periodic table is organized in such a way that elements in the same group share similar chemical properties due to their similar electron arrangements.
Electron Arrangement of Sodium
The electron arrangement of sodium can be determined by follow the Aufbau principle, which states that electrons fill the lowest energy orbitals first. For sodium, the electron arrangement is:
1s 2 2s 2 2p 6 3s 1
Let s break this down:
- 1s 2: The first two electrons fill the 1s orbital.
- 2s 2: The next two electrons fill the 2s orbital.
- 2p 6: The postdate six electrons fill the 2p orbital.
- 3s 1: The last electron goes into the 3s orbital.
This configuration shows that sodium has a single electron in its outermost shell, which is the 3s orbital. This lone electron in the outermost shell is what makes sodium highly reactive.
Significance of the Electron Arrangement of Sodium
The electron arrangement of sodium has respective important implications:
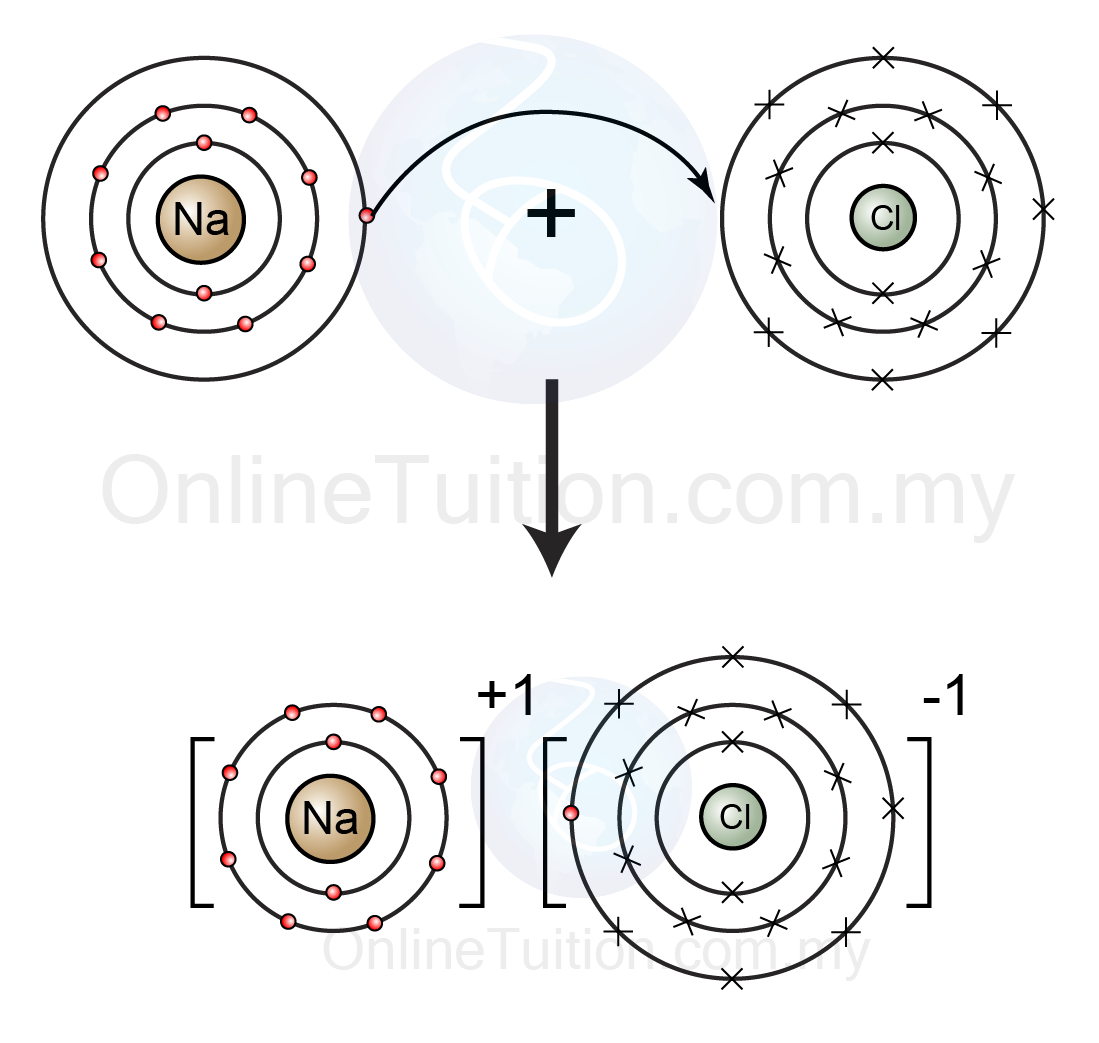
- High Reactivity: The single electron in the outermost shell makes sodium highly reactive. It pronto loses this electron to reach a stable noble gas configuration, spring a confident ion (Na ).
- Low Ionization Energy: Sodium has a low ionization energy, meaning it requires comparatively little energy to remove the outermost electron. This is why sodium is so responsive and can easily form compounds with other elements.
- Conductivity: Sodium is a good conductor of electricity and heat. This property is due to the presence of free electrons that can locomote easily through the metal.
- Chemical Bonding: Sodium s electron arrangement allows it to form ionic bonds with non metals. for instance, sodium chloride (NaCl) is formed when sodium loses an electron to chlorine, which gains an electron to complete its octet.
Chemical Properties of Sodium
The electron arrangement of sodium instantly influences its chemic properties. Some key properties include:
- Reactivity with Water: Sodium reacts violently with h2o, producing hydrogen gas and sodium hydroxide. This reaction is exothermic, intend it releases heat.
- Reactivity with Oxygen: Sodium reacts with oxygen to form sodium oxide (Na 2 O) or sodium peroxide (Na2 O2 ), depending on the conditions.
- Reactivity with Halogens: Sodium reacts with halogens (e. g., chlorine, bromine, iodine) to form sodium halides (e. g., NaCl, NaBr, NaI).
Physical Properties of Sodium
The electron arrangement of sodium also affects its physical properties. Some notable physical properties include:
- Melting and Boiling Points: Sodium has a relatively low melting point (97. 8 C) and boiling point (883 C) compared to other metals. This is due to the weak metal bonding lead from the single valency electron.
- Density: Sodium is a light metal with a concentration of 0. 97 g cm 3 at room temperature. This makes it less dense than water, although it is typically stored under oil to prevent response with air and wet.
- Appearance: Sodium is a soft, silvery white metal that can be easily cut with a knife. Its softness is due to the weak metallic bind.
Applications of Sodium
Sodium s unique properties create it worthful in diverse applications:
- Industrial Uses: Sodium is used in the production of sodium compounds, such as sodium hydroxide (NaOH) and sodium carbonate (Na 2 CO3 ), which are essential in industries like soap making, paper production, and water treatment.
- Medicinal Uses: Sodium is a crucial component of table salt (NaCl), which is essential for human health. It helps conserve fluid balance and is involved in nerve and muscle function.
- Energy Storage: Sodium is used in sodium sulfur batteries, which are eminent energy density batteries used in energy storage systems.
Safety Considerations
Due to its eminent reactivity, handling sodium requires careful precautions:
- Storage: Sodium should be stored under mineral oil to prevent contact with air and wet.
- Handling: When handling sodium, use seize personal protective equipment (PPE), include gloves and safety glasses.
- Disposal: Sodium waste should be disposed of according to local regulations to prevent environmental contamination.
Note: Always manage sodium with care to avoid accidents and ensure safety.
Comparing Sodium with Other Alkali Metals
To better interpret the electron arrangement of sodium, it s helpful to compare it with other alkali metals in Group 1. The table below shows the electron arrangements of the first few alkali metals:
| Element | Atomic Number | Electron Arrangement |
|---|---|---|
| Lithium (Li) | 3 | 1s 2 2s 1 |
| Sodium (Na) | 11 | 1s 2 2s 2 2p 6 3s 1 |
| Potassium (K) | 19 | 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1 |
| Rubidium (Rb) | 37 | 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 5s 1 |
| Cesium (Cs) | 55 | 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 5s 2 5p 6 6s 1 |
As seen in the table, each alkali metallic has a single electron in its outermost shell, which is characteristic of Group 1 elements. This similarity in electron arrangement explains why they share similar chemic properties, such as high reactivity and the tendency to form positive ions.
Conclusion
The electron arrangement of sodium is a profound aspect of its chemical and physical properties. Understanding this arrangement helps explicate sodium s high reactivity, low ionization energy, and its ability to form ionic bonds. Sodium s singular properties make it valuable in various applications, from industrial uses to medicative purposes. However, its high reactivity requires heedful handling and storage to ensure safety. By liken sodium with other alkali metals, we can see the consistency in their electron arrangements and the resulting similarities in their chemical behaviors. This knowledge is crucial for anyone canvas chemistry or work with sodium in several fields.
Related Terms:
- electron configuration for sodium
- electron dispersion diagram for sodium
- electron diagram of sodium
- electron shape diagram for sodium
- electric contour of sodium
- electron config for sodium